In the hundred year history of capsules, gelatin has always maintained the status of mainstream capsule materials because of its wide sources, stable physical and chemical properties and excellent processing properties. With the increase of people's preference for capsules, hollow capsules are more widely used in the fields of food, medicine and health products.
However, the occurrence and spread of mad cow disease and foot-and-mouth disease make people start to worry about animal derived products. The commonly used raw materials of gelatin are the bone and skin of cattle and pigs, and its risk has gradually attracted people's attention. In order to reduce the safety risk of hollow capsule raw materials, industry experts continue to research and develop appropriate plant-derived capsule materials.
In addition, with the increase of the variety of capsules, the diversity of their contents gradually makes people realize that there are compatibility problems between gelatin hollow capsules and some contents with special properties. For example, the contents containing aldehyde groups or reacting to produce aldehyde groups under certain conditions may lead to cross-linking of gelatin; The contents with strong reducibility may have Maillard reaction with gelatin; The content with strong hygroscopicity will make the shell of Ming capsule lose water and lose its original toughness. The stability of gelatin hollow capsule makes the development of new capsule materials attract more attention.
What plant derived materials are suitable for the production of hollow hard capsules? People have tried a lot. Chinese patent document application No.: 200810061238 X applied for taking cellulose sodium sulfate as the main capsule material; 200510013285.3 applied for taking starch or starch composition as the main capsule material; Wang GM [1] reported that hollow capsules were made from chitosan capsules; Zhang Xiaoju et al. [2] reported products with Konjac soybean protein as the main capsule material. Of course, the most studied materials are cellulose materials. Among them, hollow capsules made of hydroxypropyl methylcellulose (HPMC) have formed large-scale production.
HPMC is widely used in the field of food and medicine. It is a commonly used pharmaceutical excipient, which is included in Pharmacopoeia of various countries; FDA and EU approve HPMC as a direct or indirect food additive; Gras is a safe substance, No. GRN 000213; According to JECFA database, INS no.464, there is no limit on the maximum daily dose of HPMC; In 1997, it was approved by the Ministry of health of China as a food additive and thickener (No. 20), which is applicable to all kinds of food and added according to the production requirements [2-9]. Due to the difference of properties between HPMC and gelatin, the prescription of HPMC hollow capsule is more complex, and some gelling agents need to be added, such as Arabic gum, carrageenan (seaweed gum), starch, etc.
HPMC hollow capsule is a product with natural concept. Its material and production technology are recognized by Jewish, Islamic and vegetarian associations. It can meet the needs of people with various religions and eating habits, and has a high acceptance. In addition, HPMC hollow capsules have the following unique properties:
1.Low water content - about 60% lower than gelatin hollow capsule
The water content of gelatin hollow capsules is generally 12.5% - 17.5% [10]. The temperature and humidity of the environment should be controlled within an appropriate range during the production, transportation, use and preservation of hollow capsules. The appropriate temperature is 15-25 ℃ and the relative humidity is 35% - 65%, so that the performance of the product can be maintained for a long time. The water content of HPMC membrane is very low, generally 4% - 5%, which is about 60% lower than that of gelatin hollow capsule (Fig. 1). Water exchange with the environment during long-term storage will increase the water content of HPMC hollow capsule in the specified packaging, but it will not exceed 9% within 5 years.
Fig1. LOD comparison of HMPC and Gelatin shells in under different RH
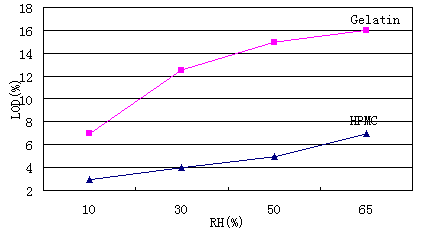
The characteristic of low water content makes HPMC hollow capsule suitable for filling hygroscopic or water sensitive contents to prolong the shelf life of the product.
2.High toughness, no brittleness
As mentioned above, the gelatin film has a specified moisture content. If it is lower than this limit, the gelatin film will be significantly brittle. Ordinary gelatin hollow capsules without any additives have a risk of brittleness of more than 10% when the moisture content is 10%; When the water content continues to decrease to 5%, 100% brittleness will occur. In contrast, the toughness of HPMC hollow capsules is much better, and they maintain good performance even if the environmental humidity is low (Fig. 3). Of course, the embrittlement rate of HPMC hollow capsules with different prescriptions under low humidity will show great differences.
On the contrary, gelatin hollow capsules placed in a high humidity environment will soften, deform or even collapse after absorbing water. HPMC hollow capsule can maintain good shape and performance even under the condition of high humidity. Therefore, HPMC hollow capsule has strong adaptability to the environment. When the sales area of the product covers a variety of climatic zones or the storage conditions are relatively poor, this advantage of HMPC hollow capsule is particularly significant.
3.Strong chemical stability
The crosslinking reaction of gelatin capsules is a thorny problem encountered by capsule preparations. Because the aldehyde group of the content reacts with the amino group of amino acids in gelatin to form a network structure, the capsule shell is difficult to dissolve under the condition of dissolution in vitro, which affects the release of drugs. Hydroxypropyl methylcellulose is a cellulose derivative, which is chemically inert and has excellent compatibility with most substances. Therefore, HPMC hollow capsule has no risk of cross-linking reaction and high chemical stability
4.Good coating performance
Enteric coated capsules are used for drugs that are easy to be damaged by gastric acid, irritating to gastric mucosa or need targeted administration. The internationally accepted technology of enteric coated capsules is the overall coating of enteric coated pellets and capsules. HPMC hollow capsule shows unique advantages in the overall coating of the capsule.
The research shows that due to the rough surface of HPMC hollow capsule, the affinity with most enteric coating materials is significantly higher than gelatin, and the adhesion speed and uniformity of coating materials are significantly better than gelatin, especially the coating reliability of body cap junction is significantly improved. In vitro dissolution test showed that the permeability of HPMC capsule in stomach was lower and there was good release in intestine[
Conclusion
The characteristics of HPMC hollow capsule have broadened its application field. From all natural products to moisture sensitive or hygroscopic contents, it also has unique applications in the field of dry powder inhalants and enteric coating. It should be noted that HPMC hollow capsules currently on the market at home and abroad have relatively high oxygen permeability and slightly slower disintegration than gelatin hollow capsules, but their bioavailability in vivo is similar [11], which should be considered in research and development.
As we all know, there is a long way to go from laboratory research, large-scale experiment, industrial production to market promotion. This is why, after years of research and development, only a few hollow capsule products made of plant-derived materials have been successfully listed. In 1997, capsugel took the lead in listing HPMC hollow capsule vcapstm in the United States, providing a new choice for oral capsule. At present, the annual sales volume of HPMC hollow capsules in the world has exceeded 20 billion, and is growing at a rate of 25% per year.
Post time: May-06-2022






